 With Angelina Jolie’s electing to have a double mastectomy because she carried the BRCa Gene, and her mother and aunt died at a very early age of the disease, the issue of genetic testing is in the forefront again. This is a three-part essay explores genetic testing when it comes to newborn testing (part one and part two) and concludes with exploring personal choices, availability, and psychological ramifications of genetic testing.
With Angelina Jolie’s electing to have a double mastectomy because she carried the BRCa Gene, and her mother and aunt died at a very early age of the disease, the issue of genetic testing is in the forefront again. This is a three-part essay explores genetic testing when it comes to newborn testing (part one and part two) and concludes with exploring personal choices, availability, and psychological ramifications of genetic testing.

After nine months of worrying and diligent pre-natal care, the day to meet your unborn child is here. Labor is long and for hours you lie in the birthing suite riding out contraction after contraction. The moment finally arrives and you discover you have a son; ten fingers, ten toes and seemingly healthylungs by the cry that you hear. He is then quickly taken over to the nurse’s station and a drop of blood from his heel is placed into a machine that in seconds will decode his entire genome. Soon your son’s future will be written in stone; he has a life expectancy of 30.2 years and has a 99% chance of dying from heart failure. In that instance, your son has been labeled as an in-valid and he is now doomed to exist within a lower class of society, one that will prohibit him from pursuing his dreams. Society has discriminated against your new baby boy based solely upon his DNA. A new form of eugenics is born.
This was the opening scene of the 1997 science-fiction movie called “Gattaca.” Besides pushing the bounds of human imagination, science fiction can serve as a warning about a future caused by the abuses of humankind. The opening birth scene in this movie is quickly becoming a potential reality. Now, a person’s entire genome can be decoded and in an instant, a person knows whether he or she will be susceptible to Parkinson’s disease, Alzheimer’s disease, Cancer, or other life threatening conditions. Proponents of genetic sequencing believe that this is the holy grail of medical care and tout phrases like “personalized medical care” and “significantly reduced costs of healthcare.” There is a rapid movement towards this goal through the proposed expansion of newborn screening for eighty-four conditions, most of which are not understood or have no known treatment.
In December of 2008, the President’s Council on Bioethics issued a report exploring the ethical issues surrounding expanded newborn screening programs. In “The Changing Moral Focus of Newborn Screening,” the Council cautions against mandating expanded  newborn screening for conditions that are not well understood and recommends staying with the Wilson-Jungner Principle for screening conditions, which has a “screen for what you can treat” philosophy that the medical community has subscribed to for the last forty years. The Council did, however, recognize an inherent public benefit to expanded newborn screening to include diseases in need of further research but stressed that this should be done only on a voluntary basis with education and follow up programs. Implementing expanded newborn screening programs with technology that produces such a high number of false positives and screening for conditions that are not well understood, on a population wide basis, is fraught with ethical implications.
newborn screening for conditions that are not well understood and recommends staying with the Wilson-Jungner Principle for screening conditions, which has a “screen for what you can treat” philosophy that the medical community has subscribed to for the last forty years. The Council did, however, recognize an inherent public benefit to expanded newborn screening to include diseases in need of further research but stressed that this should be done only on a voluntary basis with education and follow up programs. Implementing expanded newborn screening programs with technology that produces such a high number of false positives and screening for conditions that are not well understood, on a population wide basis, is fraught with ethical implications.
In this posting, I will explore the foundational test, PKU and the failures and successes. From there, I will examine the first of the four issues that relate to expanded newborn screening on a population wide basis: the best interest of the patient and the changing principle that guides screening of conditions. In the second part of this series, I will examine three additional items: 1) the examination of false positives, lack of empirical evidence, and dangers to the child and parents, 2) the determination of psychosocial ramifications of expanded newborn screening, and 3) who really benefits from expanded newborn screening – the child, parents, or the public at large. Through this exploration, one may observe how expanded newborn screening can become the launch point of genetic decoding at birth.
The Failures and Success of Screening for Phenylketonuria (PKU)
 Before these issues are explored, a refresher on Phenylketonuria, or PKU screening, may prove beneficial. Newborn screening has been around for a long time; counting ten fingers and ten toes meant your baby was healthy. In 1960, microbiologist Robert Guthrie, whose newborn son was left severely retarded from undiagnosed PKU, introduced the first genetic newborn screening test. Because of the lives that PKU screening has saved, it has been hailed as the “epitome of the application of human biochemical genetics” and the model for genetic newborn screening.
Before these issues are explored, a refresher on Phenylketonuria, or PKU screening, may prove beneficial. Newborn screening has been around for a long time; counting ten fingers and ten toes meant your baby was healthy. In 1960, microbiologist Robert Guthrie, whose newborn son was left severely retarded from undiagnosed PKU, introduced the first genetic newborn screening test. Because of the lives that PKU screening has saved, it has been hailed as the “epitome of the application of human biochemical genetics” and the model for genetic newborn screening.
Despite its successes, PKU screening had a very dark beginning, due to prematurely implementing the program to the public. PKU screening was such a disaster in its early stages it has been called the “worst test in the history of the world. “ The false positive rate for this test was about 95%, which meant that every child-testing positive had a 20 to 1 chance of not having the disease. Babies that screened positive, even false positive, were started immediately on a low phenylalanine diet. Of those that were false positive, the effects were starvation, retardation, or even death of the child. Children, numbers that are unknown even today, were made retarded by this program. The program was halted until a true positive could be distinguished from a false positive and the treatment was perfected. This took ten years. Steps that needed to be taken before implementation did not occur until after the fact.
In 2006, Norman Fost compared the proposed expansion of newborn screening to the early days of PKU screening calling it “a calamity involving every child in America that will extensively harm them and their families” because no systematic studies have been done. In fact, Fost further likens it to the premature implementation of PKU, twenty years from now, no one will know which children were helped and which were harmed from expanded newborn screening. When screening is done on asymptomatic individuals for genetic abnormalities before well-designed empirical evidence exists for the purpose of showing that screening decreases the number of newborn deaths, then it is considered an experiment.
Best Interest of the Patient and The Changing Principle for Screening
Since the period of enlightenment, western society has been on a quest for knowledge to live longer, cure diseases, and find the so-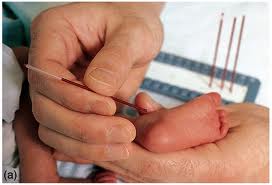 called fountain of youth. This quest dates back to the seventeenth century, when Rene Descartes, who in Discourse on Method, foresaw a new medicine that would improve the human body and mind so that we become wiser, competent, and even impervious to aging. Science has given us tools to increase our life span and treat diseases that were previously a death sentence. However, for every disease conquered new diseases emerge. In other words, disease will never be completely eradicated.
called fountain of youth. This quest dates back to the seventeenth century, when Rene Descartes, who in Discourse on Method, foresaw a new medicine that would improve the human body and mind so that we become wiser, competent, and even impervious to aging. Science has given us tools to increase our life span and treat diseases that were previously a death sentence. However, for every disease conquered new diseases emerge. In other words, disease will never be completely eradicated.
The promise of technology compounded with the resulting promise of a healthy child can make it difficult to see an ethical problem. After the anxiety and exhaustion of labor and delivery, all the parents want to know is that their child is healthy. Unless research was done before birth or there is informed consent, parents will more than likely be  ignorant of any newborn screening that occur – I know I was, because it is standard protocol to screen your newborn right after death for PKU. However, balled up into this test is so much more – a point that I will go into more depth later.
ignorant of any newborn screening that occur – I know I was, because it is standard protocol to screen your newborn right after death for PKU. However, balled up into this test is so much more – a point that I will go into more depth later.
Today, genetic testing is not only socially acceptable, but desirable. Parents rarely refused testing because of the need to know that their child was health. However, having that knowledge provides a false sense of security. Where judgment can be clouded, it is necessary that those, not associated with families, ensure the best interest of the patient is met. Just because the availability of suitable screening exists, does not mean that the disease needs to be screened for unless it is “important, relatively prevalent, and amendable to early treatment.” Unfortunately, there is a tendency for physicians to equate biomedical good with the whole of the patient’s good.
The best interest of the child does not mean genetically perfect. Rony E. Duncan believes that having knowledge of genetic abnormalities can be devastating causing the patient debilitating anxiety, depression, and despair. The American Academy of Pediatrics state that the best interest of the child is paramount. This is accomplished by promoting parent/ child understanding of relevant information, ensuring privacy and confidentiality for test results, and providing or referring children for counseling and testing when necessary. They also state that experimental tests should be offered on a voluntary basis and with informed consent, and in my opinion, the psychological ramifications attached to the potential results, false positive or true..
Within the ten criteria that the Wilson-Jungner principle outlines, a word of caution is mentioned about embracing population-wide screening before the conditions are well understood. Organizations like the National Institute of Child Health and Human Development (NICHD) have cited the “screen for what you can treat” philosophy as dogma that should be discarded” and proposed a new principle for screening- treat for what will provided “substantial public benefit.” However, this benefit is not defined or limited to the timely and effective treatment of the infant’s condition. The Committee of the National Research Council has defined “substantial public benefit” as (1) to provide management and support even when direct treatment is unavailable to the infant, (2) to inform the family of subsequent reproductive decisions and (3) to provide knowledgeof the true range and incidence of the condition to society. This definition certainly includes the societal benefits as including the opportunity for progress in biomedical research. The American College of Medical Genetics is quite explicit in embracing this broader notion of public benefit, which is not limited to the direct treatment of the child, but rather benefits the family and society about the “value of studying rare and obscure disorders in order to understand them and to find effective treatment. “
Michele Stopera Freyhauf is currently a Doctoral Student in the Department of Theology and Religionat Durham University. She has a Master of Arts Degree from John Carroll Universityin Theology and Religious Studies, is a Member of Sigma Nu, performed post-graduate work in History focusing on Gender, Religion, and Sexuality at the University of Akron, and is an Adjunct Instructor in the Religious Studies Department at Ursuline College. Her full bio is on the main contributor’s page or at http://durham.academia.edu/MSFreyhauf. Michele can be followed on twitter at @msfreyhauf.
Discover more from Feminism and Religion
Subscribe to get the latest posts sent to your email.



This is scary stuff! It makes me wonder if ignorance can be bliss, at least for a few days after birth until a mother is feeling friskier and parents are ready to learn more about their new child.
LikeLike